12th Standard Chemistry Subjects Question Paper Software Subscription
QB365 covers complete information about Tamilnadu 12th Standard 2022-2023 Chemistry Subject. Question Bank includes 12th Standard 2022-2023 Chemistry's Book back questions, other important questions, Creative questions, Extra questions, PTA quesions, Previous Year questions and other key points also. All question with detailed answers are readily available for preparting Chemistry question papers.
All Chapters Covered

Create Unlimited Question Papers

Access anywhere anytime

Multiple Pattern Question Papers
Share your Question Paper

Font size, line spacing, watermark etc,
Our Other Subjects 12th Standard
12th Standard Chemistry English Medium Chapters / Lessons 2022-2023
Metallurgy
p - Block Elements - I
p - Block Elements - II
Transition and Inner Transition Elements
Coordination Chemistry
Solid State
Chemical Kinetics
Ionic Equilibrium
Electro Chemistry
Surface Chemistry
Hydroxy Compounds and Ethers
Carbonyl Compounds and Carboxylic Acids
Organic Nitrogen Compounds
Biomolecules
Chemistry In Everyday Life
12th Standard Chemistry English Medium Chapters / Lessons 2022-2023
Metallurgy
p - Block Elements - I
p - Block Elements - II
Transition and Inner Transition Elements
Coordination Chemistry
Solid State
Chemical Kinetics
Ionic Equilibrium
Electro Chemistry
Surface Chemistry
Hydroxy Compounds and Ethers
Carbonyl Compounds and Carboxylic Acids
Organic Nitrogen Compounds
Biomolecules
Chemistry In Everyday Life
12th Standard Chemistry English Medium Chapters / Lessons 2022-2023 Syllabus
Metallurgy
Introduction - Occurrence of metals - Concentration of ores - Extraction of crude metal - Thermodynamic principle of metallurgy - Electrochemical principle of metallurgy - Refining process - Application metals -
p - Block Elements - I
Introduction - General trends in properties of p-block elements - Group 13 (Boron group) elements - Group 14 (Carbon group) elements -
p - Block Elements - II
Introduction - Group 15 (Nitrogen group) elements - oxygen - Group 17 (Halogen group) elements - Group 18 (Inert gases) elements -
Transition and Inner Transition Elements
Introduction - Position of d- block elements in the periodic table - Electronic configuration - General trend in properties - important compound of Transition elements
Coordination Chemistry
Introduction - Coordination compounds and double salts - Werner's theory of coordination compounds - Definition of important terms pertaining to co-ordination compounds - Nomenclature of coordination compounds - Isomerism in coordination compounds - Theories of coordination compound
Solid State
Introduction - General characteristics of solids - Classification of solids - Classification of crystalline solids - Classification of crystalline solids - Primitive and non-primitive unit cell - Packing in crystals - Imperfection in solids
Chemical Kinetics
Introduction - Rate of a chemical reaction - Molecularity - The integrated rate equation - Half-life period of a reaction - Collision theory - Arrhenius equation – The effect of temperature on reaction rate - Factors affecting the reaction rate
Features in Question Paper Preparation software

(or) type Question

Add or Remover

Sub Questions

Adding Notes

Multiple Pattern

All subjects available
How to Create 12th Standard Chemistry English Medium Question Paper
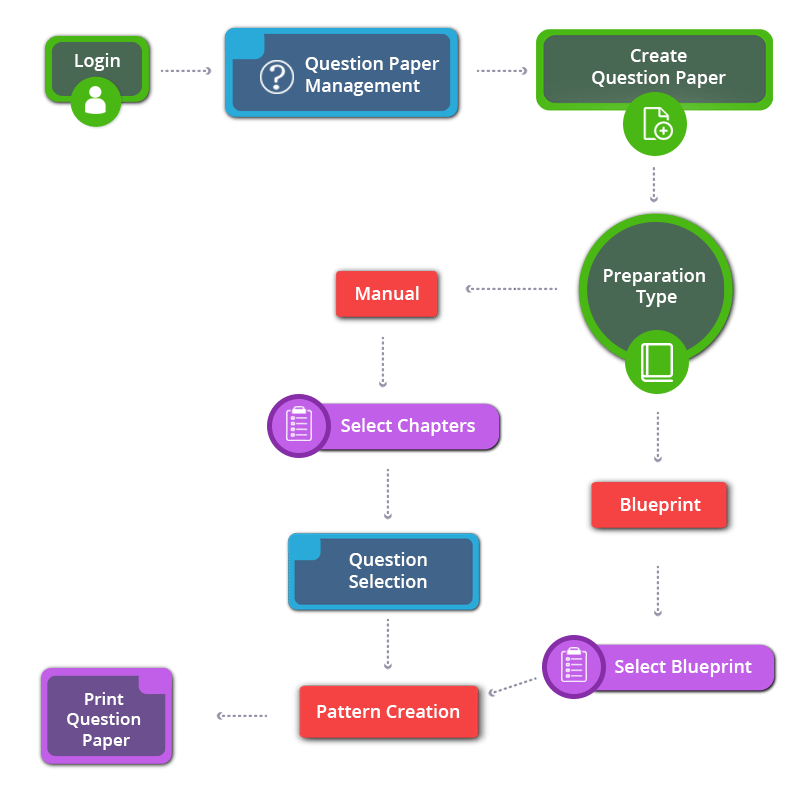

12th Standard Chemistry English Medium Chapters / Lessons 2022-2023
- Covers all chapters
- Unique Creative Questions
- Unlimited Question Paper
- Multiple Patterns & Answer keys
3267
2940



