- State Board
-
12th Standard
-

Biology
-

Computer Applications
-

Computer Science
-

Business Maths and Statistics
-

Commerce
-

Economics
-

Maths
-

Chemistry
-

Physics
-

Computer Technology
-

History
-

Accountancy
-

Tamil
-

Maths
-

Chemistry
-

Physics
-

Biology
-

Computer Science
-

Business Maths and Statistics
-

Economics
-

Commerce
-

Accountancy
-

History
-

Computer Applications
-

Computer Technology
-

English
12th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
11th Standard
-

Maths
-

Biology
-

உயிரியல் - தாவரவியல்
-

Economics
-

Physics
-

Chemistry
-

History
-

Business Maths and Statistics
-

Computer Science
-

Accountancy
-

Commerce
-

Computer Applications
-

Computer Technology
-

Tamil
-

Maths
-

Commerce
-

Economics
-

Biology
-

Business Maths and Statistics
-

Accountancy
-

Computer Science
-

Physics
-

Chemistry
-

Computer Applications
-

History
-

Computer Technology
-

Tamil
-

English
11th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
9th Standard
-

-

-

-

-

-

-

Maths
-

Science
-

Social Science
-

Maths
-

Science
-

Social Science
9th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
6th Standard
-

Maths
-

Science
-

Social Science
-

Maths
-

Science
-

Social Science
6th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
10th Standard
-

Maths
-

Science
-

Social Science
-

Tamil
-

Maths
-

Science
-

Social Science
-

English
-

English
10th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
7th Standard
-

Maths
-

Science
-

Maths
-

Science
-

Social Science
7th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
8th Standard
-

கணிதம் - old
-

Science
-

Social Science
-

கணிதம்
-

Maths
-

Science
-

Social Science
8th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
12th Standard
- CBSE Board
-
12th Standard CBSE
-

Biology
-

Physics
-

Chemistry
-

Maths
-

Accountancy
-

Introductory Micro and Macroeconomics
-

Business Studies
-

Economics
-

Computer Science
-

Geography
-

English
-

History
-

Indian Society
-

Physical Education
-

Sociology
-

Tamil
-

Bio Technology
-

Engineering Graphics
-

Entrepreneurship
-

Hindi Core
-

Hindi Elective
-

Home Science
-

Legal Studies
-

Political Science
-

Psychology
12th Standard CBSE Subject Question Paper & Study Material
-
-
11th Standard CBSE
-

Mathematics
-

Chemistry
-

Biology
-

Physics
-

Business Studies
-

Accountancy
-

Economics
-

Computer Science
-

Bio Technology
-

English
-

Enterprenership
-

Geography
-

Hindi
-

History
-

Home Science
-

Physical Education
-

Political Science
-

Psychology
-

Sociology
-

Applied Mathematics
11th Standard CBSE Subject Question Paper & Study Material
-
- 10th Standard CBSE
-
9th Standard CBSE
-

Mathematics
-

Social Science
-

Science
-

English
-

Hindi
9th Standard CBSE Subject Question Paper & Study Material
-
-
8th Standard CBSE
-

Science
-

Social Science
-

Mathematics
-

English
8th Standard CBSE Subject Question Paper & Study Material
-
-
7th Standard CBSE
-

Mathematics
-

Science
-

Social Science
-

English
7th Standard CBSE Subject Question Paper & Study Material
-
-
6th Standard CBSE
-

Mathematics
-

Science
-

Social Science
-

English
6th Standard CBSE Subject Question Paper & Study Material
-
-
12th Standard CBSE
- Free Online Test
- News
- Study Materials
-
Students
-

Stateboard Tamil Nadu
-

CBSE Board
-

Free Online Tests
-

Educational News
-

Scholarships
-

Entrance Exams India
-

Video Materials
Study Materials , News and Scholarships
-
-
Students

11th Chemistry Chapter 14 Haloalkanes and Haloarenes Book Back Questions Question Bank Software Sep-07 , 2019
Haloalkanes and Haloarenes
Haloalkanes and Haloarenes Book Back Questions
11th Standard
-
Reg.No. :
Chemistry
Time :
00:45:00 Hrs
Total Marks :
30
-
The IUPAC name of
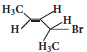 is ____________(a)
is ____________(a)2-Bromo pent – 3 – ene
(b)4-Bromo pent – 2 – ene
(c)2-Bromo pent – 4 – ene
(d)4-Bromo pent – 1 – ene
-
Of the following compounds, which has the highest boiling point?
(a)n-Butyl chloride
(b)Isobutyl chloride
(c)t-Butyl chloride
(d)n-propyl chloride
-
What should be the correct IUPAC name of diethyl chloromethane?
(a)3 – Chloro pentane
(b)1-Chloropentane
(c)1-Chloro-1, 1, diethyl methane
(d)1 –Chloro-1-ethyl propane
-
Benzene reacts with Cl2 in the presence of FeCl3 and in absence of sunlight to form ________
(a)Chlorobenzene
(b)Benzyl chloride
(c)Benzal chloride
(d)Benzene hexachloride
-
Which of the following reagent is helpful to differentiate ethylene dichloride and ethylidene chloride?
(a)Zn / methanol
(b)KOH / ethanol
(c)aqueous KOH
(d)ZnCl2 / Con HCl
-
Why chlorination of methane is not possible in dark?
-
How will you prepare n propyl iodide from n-propyl bromide?
-
Give reasons for polarity of C-X bond in halo alkane.
-
Why is it necessary to avoid even traces of moisture during the use of Grignard reagent?
-
What happens when acetyl chloride is treated with excess of CH3MgI?
-
Write a chemical reaction useful to prepare the following:
i) Freon-12 from Carbon tetrachloride
ii) Carbon tetrachloride from carbon disulphide -
Explain the mechanism of SN1 reaction by highlighting the stereochemistry behind it
-
Explain the preparation of the following compounds
i) DDT
ii) Chloroform
iii) Biphenyl
iv) Chloropicrin
v) Freon-12 -
An organic compound (A) with molecular formula C2H5Cl reacts with KOH gives compounds (B) and with alcoholic KOH gives compound (C). Identify (A),(B), and (C)
-
A hydrocarbon C3H6 (A) reacts with HBr to form compound (B). Compound (B) reacts with aqueous potassium hydroxide to give (C) of molecular formula C3H6O.what are (A) (B) and (C). Explain the reactions.
-
Two isomers (A) and (B) have the same molecular formula C2H4Cl2. Compound (A) reacts with aqueous KOH gives compound (C) of molecular formula C2H4O. Compound (B) reacts with aqueous KOH gives compound(D) of molecular formula C2H6O2. Identify (A), (B), (C) and (D).
5 x 1 = 5
8 x 2 = 16
3 x 3 = 9






 11th Standard Chemistry Syllabus
11th Standard Chemistry Syllabus  11th Standard Chemistry Study Materials
11th Standard Chemistry Study Materials 11th Standard Chemistry MCQ Practise Tests
11th Standard Chemistry MCQ Practise Tests 

Reviews & Comments about 11th Chemistry Chapter 14 Haloalkanes and Haloarenes Book Back Questions
Write your Comment