- State Board
-
12th Standard
-

Biology
-

Computer Applications
-

Computer Science
-

Business Maths and Statistics
-

Commerce
-

Economics
-

Maths
-

Chemistry
-

Physics
-

Computer Technology
-

History
-

Accountancy
-

Tamil
-

Maths
-

Chemistry
-

Physics
-

Biology
-

Computer Science
-

Business Maths and Statistics
-

Economics
-

Commerce
-

Accountancy
-

History
-

Computer Applications
-

Computer Technology
-

English
12th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
11th Standard
-

Maths
-

Biology
-

உயிரியல் - தாவரவியல்
-

Economics
-

Physics
-

Chemistry
-

History
-

Business Maths and Statistics
-

Computer Science
-

Accountancy
-

Commerce
-

Computer Applications
-

Computer Technology
-

Tamil
-

Maths
-

Commerce
-

Economics
-

Biology
-

Business Maths and Statistics
-

Accountancy
-

Computer Science
-

Physics
-

Chemistry
-

Computer Applications
-

History
-

Computer Technology
-

Tamil
-

English
11th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
9th Standard
-

-

-

-

-

-

-

Maths
-

Science
-

Social Science
-

Maths
-

Science
-

Social Science
9th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
6th Standard
-

Maths
-

Science
-

Social Science
-

Maths
-

Science
-

Social Science
6th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
10th Standard
-

Maths
-

Science
-

Social Science
-

Tamil
-

Maths
-

Science
-

Social Science
-

English
-

English
10th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
7th Standard
-

Maths
-

Science
-

Maths
-

Science
-

Social Science
7th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
8th Standard
-

கணிதம் - old
-

Science
-

Social Science
-

கணிதம்
-

Maths
-

Science
-

Social Science
8th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
12th Standard
- CBSE Board
-
12th Standard CBSE
-

Biology
-

Physics
-

Chemistry
-

Maths
-

Accountancy
-

Introductory Micro and Macroeconomics
-

Business Studies
-

Economics
-

Computer Science
-

Geography
-

English
-

History
-

Indian Society
-

Physical Education
-

Sociology
-

Tamil
-

Bio Technology
-

Engineering Graphics
-

Entrepreneurship
-

Hindi Core
-

Hindi Elective
-

Home Science
-

Legal Studies
-

Political Science
-

Psychology
12th Standard CBSE Subject Question Paper & Study Material
-
-
11th Standard CBSE
-

Mathematics
-

Chemistry
-

Biology
-

Physics
-

Business Studies
-

Accountancy
-

Economics
-

Computer Science
-

Bio Technology
-

English
-

Enterprenership
-

Geography
-

Hindi
-

History
-

Home Science
-

Physical Education
-

Political Science
-

Psychology
-

Sociology
-

Applied Mathematics
11th Standard CBSE Subject Question Paper & Study Material
-
- 10th Standard CBSE
-
9th Standard CBSE
-

Mathematics
-

Social Science
-

Science
-

English
-

Hindi
9th Standard CBSE Subject Question Paper & Study Material
-
-
8th Standard CBSE
-

Science
-

Social Science
-

Mathematics
-

English
8th Standard CBSE Subject Question Paper & Study Material
-
-
7th Standard CBSE
-

Mathematics
-

Science
-

Social Science
-

English
7th Standard CBSE Subject Question Paper & Study Material
-
-
6th Standard CBSE
-

Mathematics
-

Science
-

Social Science
-

English
6th Standard CBSE Subject Question Paper & Study Material
-
-
12th Standard CBSE
- Free Online Test
- News
- Study Materials
-
Students
-

Stateboard Tamil Nadu
-

CBSE Board
-

Free Online Tests
-

Educational News
-

Scholarships
-

Entrance Exams India
-

Video Materials
Study Materials , News and Scholarships
-
-
Students

11th Standard Chemistry English Medium - Important 3 Mark Question Paper and Answer Key 2022 - 2023 Study Materials Dec-31 , 2022
QB365 provides a detailed and simple solution for every Possible Questions in Class 11 Chemistry Subject - Important 3 Mark English Medium. It will help Students to get more practice questions, Students can Practice these question papers in addition to score best marks.
11th Standard Chemistry Important 3 Mark Question with Answers
11th Standard
-
Reg.No. :
Chemistry
Time :
02:00:00 Hrs
Total Marks :
90
-
Which contains the greatest number of moles of oxygen atoms
i) 1 mol of ethanol
ii) 1 mol of formic acid
iii) 1 mol of H2O -
How fast must a 54g tennis ball travel in order to have a de Broglie wavelength that is equal to that of a photon of green light 5400\(\overset { 0 }{ A } \) ?
-
An ion with mass number 37 possesses unit negative charge. If the ion contains 11.1% more neutrons than electrons, Find the symbol of the ion.
-
Elements a, b, c and d have the following electronic configurations:
a: 1s2, 2s2, 2p6
b: 1s2, 2s2, 2p6, 3s2, 3p1
c: 1s2, 2s2, 2p6, 3s2, 3p6
d: 1s2, 2s2, 2p1
Which elements among these will belong to the same group of periodic table. -
Write chemical equation for the following reactions.
i) reaction of hydrogen with tungsten (VI) oxide on heating.
(ii) hydrogen gas and chlorine gas. -
Complete the following chemical reactions and classify them into
(a) hydrolysis (b) redox (c) hydration reactions.
(1) KMnO4 + H2O2 ➝
(2) CrCl3 + H2O ➝
(3) CaO + H2O ➝ -
Arrange NH3, H2O and HF in the order of increasing magnitude of hydrogen bonding and explain the basis for your arrangement.
-
Write balanced chemical equation for each of the following chemical reactions.
Rubidum with oxgen gas -
A sample of gas has a volume of 8.5 dm3 at an unknown temperature. When the sample is submerged in ice water at 0 °C, its volume gets reduced to 6.37 dm3. What is its initial temperature ?
-
An unknown gas diffuses at a rate of 0.5 time that of nitrogen at the same temperature and pressure. Calculate the molar mass of the unknown gas.
-
The standard enthalpies of formation of SO2 and SO3 are -297 kJ mol-1 and -396 kJ mol-1 respectively. Calculate the standard enthalpy of reaction for the reaction: SO2 + \(\frac{1}{2}\)O2⟶SO3
-
For a chemical reaction the values of ΔH and ΔS at 300K are -10 KJ mole-1 and -20 J deg-1 mole-1 respectively. What is the value of ΔG of the reaction? Calculate the ΔG of a reaction at 600K assuming ΔH and ΔS values are constant. Predict the nature of the reaction.
-
Give the IUPAC names of the following compounds.
\({ CH }_{ 2 }-{ CH }_{ 2 }-\underset { \overset { | }{ OH } }{ CH } -CHO\) -
Give the IUPAC names of the following compounds.
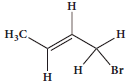
-
Give the IUPAC names of the following compounds.
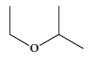
-
Give the structure for the following compound.
1,3,5- Trimethyl cyclohex - 1 -ene -
Give the structure for the following compound.
1, 3- Dimethylcyclohexane -
What are electrophiles and nucleophiles ? Give suitable examples for each.
-
Give examples for the following types of organic reactions.
Electrophilic substitution. -
Describe the mechanism of Nitration of benzene.
-
write all possible isomers for an aromatic benzenoid compound having the molecular formula C8H10
-
How will you prepare n propyl iodide from n-propyl bromide?
-
A hydrocarbon C3H6 (A) reacts with HBr to form compound (B). Compound (B) reacts with aqueous potassium hydroxide to give (C) of molecular formula C3H6O.what are (A) (B) and (C). Explain the reactions.
-
What would happen, if the greenhouse gases were totally missing in the earth’s atmosphere ?
-
How would you know whether a redox reaction is taking place in an acidic, alkaline or neutral medium?
-
How much volume of hydrogen is liberated when 0.12g of magnesium reacts with dilute hydrochloric acid ?
-
Write the name and deduce the atomic number of the following element
i. The second alkali metal
ii. The fourth noble gas
iii. The third halogen
iv. The first transition element -
Define the following terms.
-
Define the Molar Heat of Sublimation
-
What is the nature of the reaction for the following?
(i) ΔG>0
(ii) ΔG<0
(iii) ΔG=0
Answer All The Questions
30 x 3 = 90
*****************************************
Answers






 11th Standard Chemistry Syllabus
11th Standard Chemistry Syllabus  11th Standard Chemistry Study Materials
11th Standard Chemistry Study Materials 11th Standard Chemistry MCQ Practise Tests
11th Standard Chemistry MCQ Practise Tests 

Reviews & Comments about 11th Standard Chemistry English Medium - Important 3 Mark Question Paper and Answer Key 2022 - 2023
Write your Comment