- State Board
-
12th Standard
-

Biology
-

Computer Applications
-

Computer Science
-

Business Maths and Statistics
-

Commerce
-

Economics
-

Maths
-

Chemistry
-

Physics
-

Computer Technology
-

History
-

Accountancy
-

Tamil
-

Maths
-

Chemistry
-

Physics
-

Biology
-

Computer Science
-

Business Maths and Statistics
-

Economics
-

Commerce
-

Accountancy
-

History
-

Computer Applications
-

Computer Technology
-

English
12th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
11th Standard
-

Maths
-

Biology
-

உயிரியல் - தாவரவியல்
-

Economics
-

Physics
-

Chemistry
-

History
-

Business Maths and Statistics
-

Computer Science
-

Accountancy
-

Commerce
-

Computer Applications
-

Computer Technology
-

Tamil
-

Maths
-

Commerce
-

Economics
-

Biology
-

Business Maths and Statistics
-

Accountancy
-

Computer Science
-

Physics
-

Chemistry
-

Computer Applications
-

History
-

Computer Technology
-

Tamil
-

English
11th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
9th Standard
-

-

-

-

-

-

-

Maths
-

Science
-

Social Science
-

Maths
-

Science
-

Social Science
9th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
6th Standard
-

Maths
-

Science
-

Social Science
-

Maths
-

Science
-

Social Science
6th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
10th Standard
-

Maths
-

Science
-

Social Science
-

Tamil
-

Maths
-

Science
-

Social Science
-

English
-

English
10th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
7th Standard
-

Maths
-

Science
-

Maths
-

Science
-

Social Science
7th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
8th Standard
-

கணிதம் - old
-

Science
-

Social Science
-

கணிதம்
-

Maths
-

Science
-

Social Science
8th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
12th Standard
- CBSE Board
-
12th Standard CBSE
-

Biology
-

Physics
-

Chemistry
-

Maths
-

Accountancy
-

Introductory Micro and Macroeconomics
-

Business Studies
-

Economics
-

Computer Science
-

Geography
-

English
-

History
-

Indian Society
-

Physical Education
-

Sociology
-

Tamil
-

Bio Technology
-

Engineering Graphics
-

Entrepreneurship
-

Hindi Core
-

Hindi Elective
-

Home Science
-

Legal Studies
-

Political Science
-

Psychology
12th Standard CBSE Subject Question Paper & Study Material
-
-
11th Standard CBSE
-

Mathematics
-

Chemistry
-

Biology
-

Physics
-

Business Studies
-

Accountancy
-

Economics
-

Computer Science
-

Bio Technology
-

English
-

Enterprenership
-

Geography
-

Hindi
-

History
-

Home Science
-

Physical Education
-

Political Science
-

Psychology
-

Sociology
-

Applied Mathematics
11th Standard CBSE Subject Question Paper & Study Material
-
- 10th Standard CBSE
-
9th Standard CBSE
-

Mathematics
-

Social Science
-

Science
-

English
-

Hindi
9th Standard CBSE Subject Question Paper & Study Material
-
-
8th Standard CBSE
-

Science
-

Social Science
-

Mathematics
-

English
8th Standard CBSE Subject Question Paper & Study Material
-
-
7th Standard CBSE
-

Mathematics
-

Science
-

Social Science
-

English
7th Standard CBSE Subject Question Paper & Study Material
-
-
6th Standard CBSE
-

Mathematics
-

Science
-

Social Science
-

English
6th Standard CBSE Subject Question Paper & Study Material
-
-
12th Standard CBSE
- Free Online Test
- News
- Study Materials
-
Students
-

Stateboard Tamil Nadu
-

CBSE Board
-

Free Online Tests
-

Educational News
-

Scholarships
-

Entrance Exams India
-

Video Materials
Study Materials , News and Scholarships
-
-
Students
+1 FIRST REVISION TEST
11th Standard
-
Reg.No. :
Chemistry
Time :
02:30:00 Hrs
Total Marks :
70
-
7.5 g of a gas occupies a volume of 5.6 litres at 0° C and 1 atm pressure. The gas is ________.
(a)NO
(b)N2O
(c)CO
(d)CO2
-
Which of the following provides the experimental justification of magnetic quantum number?
(a)Zeeman effect
(b)Stark effect
(c)Uncertainty principle
(d)Quantum condition
-
Which one of the following is true about metallic character when we move from left to right in a period and top to bottom in a group?
(a)Decreases in a period and increases along the group
(b)Increases in a period and decreases in a group
(c)Increases both in the period and the group
(d)Decreases both in the period and in the group
-
Which pair are not hydrogen isotopes?
(a)Ortho and para hydrogen
(b)Protium and deuterium
(c)Deuterium and tritium
(d)Tritium and protium
-
Which of the following has the highest tendency to give the reaction \(M_{g}^{+}\xrightarrow[Medium]{Aqueous}M_{aq}^{+}\)
(a)Na
(b)Li
(c)Rb
(d)K
-
Identify the correct mathematical expression of Graham's law of diffusion __________
(a)\(\frac { { r }_{ 2 } }{ { r }_{ 1 } } =\sqrt { \frac { { M }_{ 2 } }{ { M }_{ 1 } } } \)
(b)\({ r }_{ 1 }r_{ 2 }=\sqrt { \frac { { M }_{ 2 } }{ { M }_{ 1 } } } \)
(c)\(\frac { { r }_{ 1 } }{ { r }_{ 2 } } =\sqrt { \frac { { M }_{ 2 } }{ { M }_{ 1 } } } \)
(d)\(\frac { { r }_{ 1 } }{ { r }_{ 2 } } ={ \left[ \frac { { M }_{ 2 } }{ { M }_{ 1 } } \right] }^{ 2 }\)
-
If one mole of ammonia and one mole of hydrogen chloride are mixed in a closed container to form ammonium chloride gas, then _____________
(a)ΔH > ΔU
(b)ΔH - ΔU = 0
(c)ΔH + ΔU = 0
(d)ΔH < ΔU
-
For the homogeneous gas reaction at 600K \(4NH_{ 3\left( g \right) }+5{ O }_{ 2\left( g \right) }\rightleftharpoons 4NO_{ \left( g \right) }+6{ H }_{ 2 }{ O }_{ \left( g \right) }\) The unit of equilibrium constant K" is
(a)(mol dm-3)-1
(b)(mol dm-3)
(c)(mol dm-3)10
(d)(mol dm-3)9
-
Normality of 1.25M sulphuric acid is ___________
(a)1.25 N
(b)3.75 N
(c)2.5 N
(d)2.25 N
-
During change of \({ O }_{ 2 }\) to \({ O }_{ 2 }^{ - }\) ion, the electron adds on which one of the following orbitals?
(a)\(\pi\) orbitals
(b)\(\sigma -\)orbitals
(c)\(\pi^*\) orbitals
(d)\(\sigma ^*\)orbitals
-
The IUPAC name of the Compound is _____________
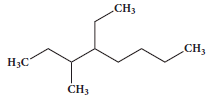 (a)
(a)2,3 - Diemethylheptane
(b)3- Methyl -4- ethyloctane
(c)5-ethyl -6-methyloctane
(d)4-Ethyl -3 - methyloctane
-
The most stable carbocation is ____________
(a)\(C{ H }_{ 3 }-\overset { + }{ C } { H }_{ 2 }\)
(b)\(C{ H }_{ 3 }-\overset { + }{ C } H-C{ H }_{ 3 }\)
(c)\(C{ H }_{ 2 }=CH-\overset { + }{ C } { H }_{ 2 }\)
(d)\(\overset { + }{ C } { H }_{ 3 }\)
-
In which of the following molecules, all atoms are co-planar ___________
(a) (b)
(b) (c)
(c) (d)
(d)both (a) and (b)
-
Ozone depletion will cause ___________.
(a)forest fires
(b)eutrophication
(c)bio magnification
(d)global warming
-
Cause of eutrophication is ____________
(a)Increase of oxygen in water bodies
(b)Increase in number of aquatic organisms
(c)Nutrient enrichment of water bodies
(d)All ofthese
-
Calculate the equivalent masses of the following - Oxalic acid H2C2O4
-
Magnesium loses electrons successively to form Mg+, Mg2+ and Mg3+ ions. Which step will have the highest ionisation energy and why?
-
Lithium iodide is covalent. Explain why?
-
Define the calorific value of food. What is the unit of calorific value?
-
What are the non-aqueous solution ? Give example
-
Give a brief description of the principles of
Fractional distillation -
Which of the following ions is more stable ? Use resonance to explain your answer.
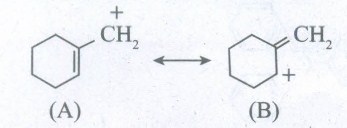
-
Identify A, Band C from the following equation
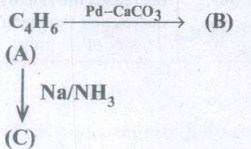
-
What is green chemistry ?
-
Calculate the uncertainty in position of an electron, if Δv = 0.1% and \(\upsilon \) = 2.2 x 106 ms-1.
-
Mention any two biological effects of D2O.
-
Do you think that heavy water can be used for drinking purposes?
-
Give the systematic names for the following
Milk of magnesia -
Which of the following gases would you expect to deviate from ideal behaviour under conditions of low temperature F2, Cl2 or Br2? Explain.
-
For the reaction
SrCO3 (s) ⇌ SrO (s) + CO2(g),
the value of equilibrium constant KP = 2.2 x 10–4 at 1002 K. Calculate KC for the reaction. -
What happens when the' concentration of H2 and I2 are increased in the reaction \({ H }_{ 2 }+{ I }_{ 2 }\rightleftharpoons 2HI?\)
-
0.40 g of an iodo-substituted organic compound gave 0.235 g of AgI by carius method. Calculate the percentage of iodine in the compound. (Ag = 108, I = 127)
-
Carry over the following reaction mechanisms.
(i) Bromination of alkene
(ii) Addition of HCN to CH3CHO
(iii) Formation of alkyl bromide with benzoyl peroxide as radical initiator. -
A Compound on analysis gave Na = 14.31% S = 9.97% H = 6.22% and 0 = 69.5%.
Calculate the molecular formula of the compound if all the hydrogen in the compound is present in combination with oxygen as a water of crystallization. (molecular mass of the compound is 322). -
The Li2+ ion is a hydrogen like ion that can be described by the Bohr model. Calculate the Bohr radius of the third orbit and calculate the energy of an electron in 4th orbit.
-
List out and compare the chemical properties of metals and non-metals.
-
Define hydrogen bond and its types.
-
Alkaline earth metal (A), belongs to 3rd period reacts with oxygen and nitrogen to form compound (B) and (C) respectively. It undergo metal displacement reaction with AgNO3 solution to form compound (D).
-
Consider the following reaction
Fe3+(aq) + SCN–(aq) ⇌ [Fe(SCN)]2+(aq)
A solution is made with initial Fe3+, SCN- concentration of 1 x 10-3 M and 8 x 10-4 M respectively. At equilibrium [Fe(SCN)]2+ concentration is 2 x 10-4 M. Calculate the value of equilibrium constant. -
What is the molarity of the solutions prepared by diluting 25.0 mL of 0.312M MgCl2 solution to each of the following volumes (a) 40 mL
(b) 100mL
(c) 350mL? -
How is nitrogen estimated by Dumas method?
-
Predict the product:
(i) Chloroform + O2 ⟶?
(ii) CCl4 + H2O ⟶?
(iv)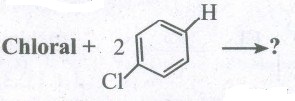
(iv) \({ CH }_{ 3 }-CH{ Cl }_{ 2 }+KOH\overset { { C }_{ 2 }{ H }_{ 5 }{ OH }_{ 3 } }{ \longrightarrow } \)?
(v) Ethylene glycol + 2PCI5 ⟶? -
How is acid rain formed ? Explain its effect
PART-A
15 x 1 = 15
PART -B
6 x 2 = 16
PART -C
6 x 3 = 18
PART -D
5 x 5 = 25






 11th Standard Chemistry Syllabus
11th Standard Chemistry Syllabus  11th Standard Chemistry Study Materials
11th Standard Chemistry Study Materials 11th Standard Chemistry MCQ Practise Tests
11th Standard Chemistry MCQ Practise Tests 

Reviews & Comments about 11th Standard Chemistry Model Question Paper 2019
Write your Comment