- State Board
-
12th Standard
-

Biology
-

Computer Applications
-

Computer Science
-

Business Maths and Statistics
-

Commerce
-

Economics
-

Maths
-

Chemistry
-

Physics
-

Computer Technology
-

History
-

Accountancy
-

Tamil
-

Maths
-

Chemistry
-

Physics
-

Biology
-

Computer Science
-

Business Maths and Statistics
-

Economics
-

Commerce
-

Accountancy
-

History
-

Computer Applications
-

Computer Technology
-

English
12th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
11th Standard
-

Maths
-

Biology
-

உயிரியல் - தாவரவியல்
-

Economics
-

Physics
-

Chemistry
-

History
-

Business Maths and Statistics
-

Computer Science
-

Accountancy
-

Commerce
-

Computer Applications
-

Computer Technology
-

Tamil
-

Maths
-

Commerce
-

Economics
-

Biology
-

Business Maths and Statistics
-

Accountancy
-

Computer Science
-

Physics
-

Chemistry
-

Computer Applications
-

History
-

Computer Technology
-

Tamil
-

English
11th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
9th Standard
-

-

-

-

-

-

-

Maths
-

Science
-

Social Science
-

Maths
-

Science
-

Social Science
9th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
6th Standard
-

Maths
-

Science
-

Social Science
-

Maths
-

Science
-

Social Science
6th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
10th Standard
-

Maths
-

Science
-

Social Science
-

Tamil
-

Maths
-

Science
-

Social Science
-

English
-

English
10th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
7th Standard
-

Maths
-

Science
-

Maths
-

Science
-

Social Science
7th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
8th Standard
-

கணிதம் - old
-

Science
-

Social Science
-

கணிதம்
-

Maths
-

Science
-

Social Science
8th Standard stateboard question papers & Study material
தமிழ் Subjects
English Subjects
-
-
12th Standard
- CBSE Board
-
12th Standard CBSE
-

Biology
-

Physics
-

Chemistry
-

Maths
-

Accountancy
-

Introductory Micro and Macroeconomics
-

Business Studies
-

Economics
-

Computer Science
-

Geography
-

English
-

History
-

Indian Society
-

Physical Education
-

Sociology
-

Tamil
-

Bio Technology
-

Engineering Graphics
-

Entrepreneurship
-

Hindi Core
-

Hindi Elective
-

Home Science
-

Legal Studies
-

Political Science
-

Psychology
12th Standard CBSE Subject Question Paper & Study Material
-
-
11th Standard CBSE
-

Mathematics
-

Chemistry
-

Biology
-

Physics
-

Business Studies
-

Accountancy
-

Economics
-

Computer Science
-

Bio Technology
-

English
-

Enterprenership
-

Geography
-

Hindi
-

History
-

Home Science
-

Physical Education
-

Political Science
-

Psychology
-

Sociology
-

Applied Mathematics
11th Standard CBSE Subject Question Paper & Study Material
-
- 10th Standard CBSE
-
9th Standard CBSE
-

Mathematics
-

Social Science
-

Science
-

English
-

Hindi
9th Standard CBSE Subject Question Paper & Study Material
-
-
8th Standard CBSE
-

Science
-

Social Science
-

Mathematics
-

English
8th Standard CBSE Subject Question Paper & Study Material
-
-
7th Standard CBSE
-

Mathematics
-

Science
-

Social Science
-

English
7th Standard CBSE Subject Question Paper & Study Material
-
-
6th Standard CBSE
-

Mathematics
-

Science
-

Social Science
-

English
6th Standard CBSE Subject Question Paper & Study Material
-
-
12th Standard CBSE
- Free Online Test
- News
- Study Materials
-
Students
-

Stateboard Tamil Nadu
-

CBSE Board
-

Free Online Tests
-

Educational News
-

Scholarships
-

Entrance Exams India
-

Video Materials
Study Materials , News and Scholarships
-
-
Students

12th Chemistry - Hydroxy Compounds and Ethers Model Question Paper Question Bank Software Jan-03 , 2020
Hydroxy Compounds and Ethers
Hydroxy Compounds and Ethers Model Question Paper
12th Standard
-
Reg.No. :
Chemistry
Time :
01:00:00 Hrs
Total Marks :
50
-
Which one of the following is the strongest acid.
(a)2 - nitrophenol
(b)4 – chlorophenol
(c)4– nitrophenol
(d)3 – nitrophenol
-
(CH3)3-C-CH(OH) CH3 \(\overset { con{ H }_{ 2 }{ SO }_{ 4 } }{ \longrightarrow } \)X (major product)
(a)(CH3)3 CCH = CH2
(b)(CH3)2C = C (CH3)2
(c)CH2= C(CH3)CH2-CH2- CH3
(d)CH2= C (CH3) - CH2- CH2- CH3
-
Assertion: Phenol is more reactive than benzene towards electrophilic substitution reaction
Reason: In the case of phenol, the intermediate arenium ion is more stabilized by resonance
Codes:
a) if both assertion and reason are true and reason is the correct explanation of assertion.
b) if both assertion and reason are true but reason is not the correct explanation of assertion.
c) assertion is true but reason is false
d) both assertion and reason are false. -
One mole of an organic compound (A) with the formula C3H8O reacts completely with two moles of HI to form X and Y. When Y is boiled with aqueous alkali it forms Z. Z answers the iodoform test. The compound (A) is ______.
(a)propan – 2-ol
(b)propan -1-ol
(c)ethoxy ethane
(d)methoxy ehane
-
Pick out the odd one among the following.
(a)CH3 - CH - OH - CH3
(b)\({ CH }_{ 3 }{ CH }_{ 2 }-\underset { \overset { | }{ { CH }_{ 3 } } }{ CH } -OH\)
(c)(CH3)3 - C - OH
(d)C6H5 - CH - OH - C2H5
-
The above reaction is called ______.(a)Gattermann reaction
(b)Williamson ether synthesis
(c)Swern's reaction
(d)Riemer tiemann reaction
-
In the sequence of reactions Identify 'C'.
(a)(b)(c)(d) -
Predict the structure of propane-1,2 diol ______.
(a)CH2 (OH) - CH2CH2OH
(b)HOCH2 - CH2OH
(c)CH3CH (OH) CH2OH
(d)None of these
-
Which of the following has an offensive odour?
(a)Phenol
(b)Benzy lalcohol
(c)Acrolein
(d)Benzyl benzoate
-
Lower alcohols like ethanol and methanol are miscible in water due to _______.
(a)their acidic character
(b)vanderwaals force of attraction
(c)dipole-dipole inter action
(d)inter molecular hydrogen bonding
-
CH2 = CH2+H2O
The above reaction is _______.
(a)Lucas test
(b)Saponification
(c)Victor Meyer's test
(d)hydroxylation
-
In the following sequence of reaction, the end product 'B' is ________.
(a)C6H5N2CI
(b)(c)(d)C6H5OH
-
Identify the major product in the given reaction
(a)(b)(c)(d) -
Intermolecular hydrogen bonds are not _______ present in.
(a)CH3COOH
(b)C2H5OC2H5
(c)CH3CH2OH
(d)C2H5NH2
-
(a) Diethylether with chlorine in presence of sunlight forms (C2Cl5)2O.
(b) The formula of diethyl oxonium chloride is (C2H2)2-O+Cl-.
(c) In anisole oxygen is strongly bonded to benzene ring.
(d) Ether is used as solvent for Grignard reagent -
Assertion: Ethanol is a weaker acid than phenol.
Reason: Sodium ethoxide may be prepared by the reaction of ethanol with aqueous NaOH.
Codes:
a) (A) and (R) are true and (R) is the correct explanation of (A)
b) Both (A) and (R) are true but (R) does not explain (A)
c) (A) is true but (R) is false
d) Both (A) and (R) are false -
Assertion: Compounds with two hydroxy groups are gem diols.
Reason: Ethanol which contains 5% methanol is known as methylated spirit.
Codes:
a) (A) and (R) are true and (R) is the correct explanation of (A)
b) Both (A) and (R) are true but (R) does not explain (A)
c) (A) is true but (R) is false
d) Both (A) and (R) are false -
Pick out the wrong statement regarding ethers.
a) dimethyl ether and diethyl ehters are simple ethers
b) ethyl methyl ether is a symmetrical ether
c) the reaction between sodium ethoxide and methyl bromide is known as williamson's ether synthesis
d) Diethyl ether is used as an anaesthetiz. -
What is the major product obtained when two moles of ethyl magnesium bromide is treated with methyl benzoate followed by acid hydrolysis.
-
Explain 'esterification' reaction with an example.
-
What happens when phenol is treated with diazonium chloride in presence of NaOH?
-
State Saytzeff's rule.
-
How is phenol prepared from
i) chloro benzene
ii) isopropyl benzene -
Complete the following sequence of reaction and Identify A, B and C.
-
Complete the following reactions
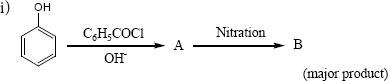
ii) \(C_6H_5-CH_{2}CH(OH)CH(CH_3)_2 \overset{ConH_2SO_4}\longrightarrow\) -
An organic compound (A) (C6H6O) gives maximum of two isomers (B) and (C) When an alkaline solution of (A) is refluxed with chloroform. (B) on oxidation gives acid (D). The acid (D) is also obtained by I treating sodium salt of (A) with CO2 under pressure followed by hydrolysis. Identify the compounds (A), (B), (C) and (D) and explain with proper chemical reactions.
-
An organic compound (A) of molecular formula C6H6O gives violet colour with neutral FeCI3. (A) gives maximum of two isomers (B) and (C) when an alkaline solution of (A) is refluxed with CCI4 (A) also reacts with C6H5N2CI to give the compound (D) which is red orange dye. Identify (A), (B), (C) and (D). Explain with suitable chemical reactions.
14 x 1 = 14
1 x 1 = 1
2 x 2 = 4
1 x 2 = 2
4 x 2 = 8
2 x 3 = 6
3 x 5 = 15






 12th Standard Chemistry Syllabus
12th Standard Chemistry Syllabus  12th Standard Chemistry Study Materials
12th Standard Chemistry Study Materials 12th Standard Chemistry MCQ Practise Tests
12th Standard Chemistry MCQ Practise Tests 

Reviews & Comments about 12th Chemistry - Hydroxy Compounds and Ethers Model Question Paper
Write your Comment